炎症性肠病(专业版)
炎症性肠病(IBD)表现为肠道肿胀和刺激症状,是由于肠腔和肠内壁的免疫失衡造成的。IBD是终生性疾病,并可导致严重腹泻、腹痛、疲劳和体重下降。
其他名称:炎症性肠炎
英文名称:Inflammatory Bowel Disease
IBD主要分为有2种类型:
下列因素可增加罹患IBD几率:
常见症状包括:
此外,溃疡性结肠炎和克罗恩病由可能发展出不同的并发症,详细可参考本网有关专文。
选项可包括如下:
调整饮食与生活方式
营养与草本综合干预
以下是基于循证医学和循证营养学有关文献综合的结果。
有助于防控炎症性肠病的营养和草本补充剂,主要包括如下:
1.益生菌:
消化道内微生物种群的变化能够局部和系统地改变免疫细胞的功能。一项研究描述了一种新型益生菌,其可以直接产生白细胞介素-10(IL-10),这是一种促进免疫耐受的抗炎细胞因子1-3。此外,摄入益生菌可以通过各种机制削弱致病菌的作用,包括竞争上皮受体结合和增强肠道屏障功能1,4,5。一些益生菌还产生丁酸,一种对结肠壁内细胞健康很重要的短链脂肪酸(见下文)6。
益生菌在IBD人群中使用的临床试验表明了有益的效果。试验的持续时间和使用的生物体各不相同,但有几次显示积极的结果7。2011年一项使用益生菌(短双歧杆菌)和益生元(低聚半乳糖)的试验表明,溃疡性结肠炎患者的临床状况显著改善8。克罗恩病的临床试验表明,每天供应500亿个或更多的益生菌生物体补充剂(如ACTIAL益生菌VSL#3)改善了肠道健康9,10。在一项试验中,两名受试者的缓解效果非常好,他们能够停止糖皮质激素药物治疗9。其他研究表明,益生菌可能抑制结直肠癌癌症发展的可能性,这是IBD患者的主要关注问题11。
另一种在IBD中显示出前景的生物是布拉氏酵母。几项试验已经证明布拉氏酵母菌对改善感染性腹泻和其他胃肠道问题的疗效12。此外,与IBD特别相关的布拉氏酵母菌似乎可以调节肠上皮的炎症反应,降低TNF-α和IL-613。同一项研究表明,布拉氏酵母促进IBD患者细胞样本中的肠道组织修复和免疫耐受。在一项随机、安慰剂对照的临床试验中,布拉氏酵母菌在常规治疗中添加后,降低了克罗恩病患者的肠道通透性14。在各种病理状态下,补充布拉氏酵母菌通常是安全有效的15。
2. 欧米伽3脂肪酸:
两种最主要的ω-3脂肪酸即二十碳五烯酸(EPA)和二十二碳六烯酸(DHA),存在于冷水鱼中16。Omega-3脂肪酸是强大的免疫调节剂,可减少循环炎症细胞因子并降低自然杀伤细胞的细胞毒性17-20。此外,在一项动物研究中,α-亚麻酸(一种植物衍生的ω-3脂肪酸)抑制了粘附分子的表达,粘附分子在炎症、免疫反应和细胞内信号事件中很重要21,22。
在临床试验中,补充鱼油可以改善克罗恩病和溃疡性结肠炎患者的脂肪酸状况,并与较低水平的炎症介质有关23-25。这些变化与疾病发作的缓解有一定的相关性26,27。鱼油还可以减少缓解所需的糖皮质激素药物的剂量27。在一项针对克罗恩病患者的研究中发现,肠溶鱼油制剂有助于降低复发率28。
现代饮食导致多数人血液中ω-6和ω-3的比例不健康,这种失衡与炎症性疾病密切相关29。一般建议将ω-6与ω-3的比例保持在4:1以下,以获得最佳健康30;这对于IBD患者可能特别重要。
3. 维生素D:
维生素D是另一种强大的免疫调节剂。实验模型表明,T细胞表达维生素D受体,缺乏维生素D信号会导致T细胞产生更高水平的炎症细胞因子。此外,维生素D是Treg细胞亚群发育所必需的,Treg细胞在抑制肠道炎症方面具有重要作用31,32。IBD患者的维生素D水平通常不足,如血清25-羟基维生素D水平较低所示33。许多其他证据也将维生素D水平低与IBD联系起来34,35。
在37名克罗恩病缓解期患者中,服用25-羟基维生素D3或骨化三醇(活性维生素D3,处方药)降低了炎症指标,改善了骨骼健康36。在一项涉及94名克罗恩病缓解期患者的双盲试验中,与安慰剂相比,每天服用1200IU维生素D3显示出复发率较低的趋势(从29%降至13%[P=0.06])37。此外,骨丢失是IBD患者的主要担忧,这种疾病以及用于治疗它的糖皮质激素都会导致骨骼健康状况不佳。补充维生素D已被证明可以在克罗恩病中保持骨密度38。
通常建议将血清25-羟基维生素D水平维持在50-80ng/mL的范围内。补充维生素D的人应定期进行25-羟基维生素D血液测试,以确保其保持在最佳范围内。
4. 维生素抗氧化剂:
抗氧化剂。正常消化会产生大量活性氧和氮物质(也称为自由基),肠粘膜对其保持广泛的抗氧化剂防御系统。然而,当出现过度氧化应激时,粘膜屏障会持续受损并渗漏,从而为炎症奠定基础39,40。此外,炎症本身会产生大量的反应性物种,破坏性的循环可能会持续下去。在患有IBD的患者中,肠道中存在高水平的活性氧,这会导致疾病造成的损害39。
在一项研究中,IBD患者的抗氧化能力明显低于非IBD患者41。一些研究表明,维生素A、维生素C、维生素E和硒的抗氧化剂组合与鱼油结合可以降低克罗恩病的某些炎症标志物42,43。此外,IBD患者血液中的类胡萝卜素和维生素C水平明显较低44。
5. 噬菌体:
噬菌体(Bacteriophages)是以细菌为目标的病毒。它们是地球上最丰富的生物,人类消化道估计含有1015个噬菌体45-47。肠道噬菌体似乎在肠道微生物群落的生态学中发挥着重要作用,包括将病毒遗传物质注入到特定细菌中,在某些情况下导致其快速死亡46-48。尽管噬菌体数量丰富,但直到最近,它们还相对较少受到研究关注46,48。新出现的证据表明噬菌体在治疗炎症性肠病方面具有治疗潜力46,45。
噬菌体似乎可以调节免疫活性,并可能影响肠道内壁的炎症。在患有炎症性肠病的个体中已经注意到噬菌体群体的疾病特异性模式,并且与在健康个体中观察到的噬菌体群体有显著差异49,50;此外,在一项研究中发现,某些噬菌体的丰度与某些类型细菌水平的降低有关49。
噬菌体疗法可能比抗生素治疗更安全,部分原因是它对正常肠道菌群的破坏最小51。在一项安全性研究中,口服补充针对肠道细菌大肠杆菌的噬菌体导致健康志愿者的粪便中可检测到这些噬菌体的存在,但在补充结束后的一周内不再检测到它们的存在。非致病性大肠杆菌的数量保持不变,没有发现不良副作用52。同样,在一项针对健康成年人的研究中,服用九种大肠杆菌靶向噬菌体的混合物,在噬菌体给药后立即收集的粪便样本中未检测到大肠杆菌,在血液、肝脏和肾脏测试中也未报告或检测到副作用53。
6.姜黄素:
姜黄素作为抗炎剂在各种环境中的功效已得到充分证明。其多重作用中最突出的是抑制核因子κB(NF-kB)信号传导。NF-kB是一种信号蛋白,它驱动无数炎症细胞因子的产生,包括白细胞介素1b(IL-1b)和白细胞介素-6(IL-6)。由于NF-kB和相关细胞因子是IBD病理学的核心,姜黄素已被研究作为一种干预措施54。
在一项研究中,姜黄素有助于减轻一小群患者的克罗恩病和溃疡性结肠炎症状,其中许多患者能够停用氨基水杨酸盐和/或糖皮质激素54,55。在一组82名溃疡性结肠炎患者中,与安慰剂加氨基水杨酸盐相比,姜黄素联合氨基水杨酸盐降低了急性发作的复发和症状严重程度。姜黄素组在治疗的6个月内复发率为4.6%,而对照组的复发率超过20%56。
7.乳香:
乳香树脂含有一种强效抗炎化合物即乙酰基-11-keto-β-boswellic acid(AKBA),简称乳香酸。一项双盲临床试验发现,在改善克罗恩病症状方面,乳香与美沙拉秦一样有效,副作用要少得多57。一项试验还发现,在30名患者中,乳香与柳氮磺胺吡啶一样有效,可以诱导溃疡性结肠炎的缓解58。这证实了早期关于乳香治疗溃疡性结肠炎患者的疗效报告59。
然而,另一项涉及108名克罗恩病患者的双盲试验并未发现乳香在维持病情缓解方面优于安慰剂60。一种名为AprèsFlex™的改良提取物(或Aflapin®),它将AKBA与其他非挥发性的乳香油结合在一起,与标准化为相同百分比的AKBA的其他制剂相比,在较低浓度下显示出改善的抗炎活性61。
8.丁酸:
丁酸(或丁酸盐)是一种短链脂肪酸,由肠道中的纤维被某些细菌代谢时产生。实验模型表明,口服丁酸盐可改善溃疡性结肠炎的炎症62。丁酸可能发挥作用的一种机制是抑制促炎细胞信号传导成分核因子κB(NF-kB)的激活63。在临床试验中,口服丁酸盐对克罗恩病和溃疡性结肠炎都有缓解作用64,65。在一项试验中,近70%的克罗恩病受试者对每天4g丁酸肠溶片的剂量有反应,持续8周。在这些应答者中,53%的应答者病情缓解,他们的NF-kB和另一种炎症因子IL-1b水平显著下降65。
9.芦荟(凝胶):
芦荟叶内部的粘液凝胶多年来一直被用于治疗溃疡性结肠炎。一项双盲随机试验发现,每天两次3盎司(约85g)的芦荟凝胶比安慰剂更好地结束了溃疡性结肠炎患者的急性发作,没有不良反应66。芦荟凝胶的免疫调节、肠道愈合和抗炎特性都可能在其功效中发挥作用67。
10.艾蒿:
艾蒿(Wormwood,又称艾草或苦艾)是一种原产于地中海地区的苦味草本植物,其标准化提取物已在克罗恩病患者身上进行了研究。与安慰剂相比,它在逐渐减少药物治疗的患者中更有效地维持病情缓解68。这可能是因为艾草可以阻断TNF-α,一种强效的促炎细胞因子69。
11. 硒:
硒是一种微量元素,对许多硒依赖性酶的功能至关重要。硒缺乏在IBD患者中很常见70-72。补充有助于缓解这一问题,这既基于血清硒的增加,也基于谷胱甘肽过氧化物酶功能的改善73。
12. L-肉碱:
L肉碱是正常细胞代谢所必需的,而肉碱水平不足尤其会影响需要大量能量的细胞,如免疫系统细胞。几项实验表明,肉碱调节炎症介质的产生,并且肉碱水平不足与炎症细胞因子的大量产生有关74,75。事实上,在一项涉及36名透析患者的临床试验中,每天补充1g L-肉碱可使CRP水平降低29%,IL-6水平降低61%76。
就肠道而言,在动物模型中,L-肉碱显著减弱了肠道组织缺氧和恢复的炎症反应77。在一项涉及121名溃疡性结肠炎受试者的随机安慰剂对照试验中,每天1或2g的丙酰-L-肉碱与安慰剂相比,在常规治疗中的缓解率更高78。在每天服用1g L肉碱组中,病情缓解率为55%,而安慰剂组仅为35%。
13.谷氨酰胺:
这是一种条件必需氨基酸,也是肠细胞(肠道吸收细胞)的主要燃料。口服谷氨酰胺补充可以稳定肠道通透性和粘膜完整性79。一项研究表明,谷氨酰胺有助于改善结肠炎动物结肠发炎部分的毛细血管血流量80。此外,中度至重度克罗恩病患者的谷氨酰胺水平较低81。
在一项随机临床试验中,每天0.5g/kg体重的谷氨酰胺剂量持续2个月,可降低克罗恩病患者的肠道通透性并改善其形态82。然而,补充谷氨酰胺的临床益处可能仅限于缓解期,因为另一项试验发现,在疾病发作期间补充谷氨酰胺并不能改善肠道通透性83。
14. 维生素K:
IBD患者经常缺乏维生素K。一项研究表明,31%的溃疡性结肠炎或克罗恩病患者缺乏维生素K84。在一项研究中,维生素K活性低与克罗恩病活性高有关85。IBD患者的维生素K缺乏也与骨密度降低有关85,86。
15. 膳食纤维:
摄入更多的膳食纤维与克罗恩病的发病率较低有关87,而摄入更多的糖与风险增加有关88。低精制糖和高膳食纤维的饮食已被证明对克罗恩病的病程有良好的影响,并且与正常饮食相比不会导致肠梗阻89。
肠道细菌发酵膳食纤维是短链脂肪酸(如丁酸)的主要来源,各种研究表明,植物纤维有助于预防溃疡性结肠炎发作90。
16. 褪黑素:
虽然褪黑素是一种有助于同步睡眠-觉醒周期的激素,但它在消化道中的产生量远大于大脑91。褪黑素降低TNF-α水平92。许多体外和动物研究表明,褪黑素可以减轻IBD的炎症93。IBD患者的褪黑激素合成增加,较高的水平与较低的症状有关,这表明这是身体减少过度炎症的努力的一部分94。
在一项针对60名接受美沙拉秦治疗的溃疡性结肠炎患者的双盲试验中,一半患者随机服用褪黑素,一半患者服用安慰剂,为期一年95。安慰剂组的炎症和临床症状有所上升,而褪黑素组仍处于缓解状态。这证实了早期一项未经控制的研究,该研究表明褪黑素对克罗恩病和溃疡性结肠炎患者有帮助96。尽管如此,还是要谨慎——至少有一项案例研究发表,其中褪黑素导致溃疡性结肠炎发作,而对糖皮质激素没有反应97。
17. 脱氢表雄酮(DHEA):
DHEA在预防慢性炎症和维持健康免疫功能方面发挥着重要作用。已发表的研究将低水平的DHEA与慢性炎症联系起来,并且DHEA已被证明可以抑制促炎细胞因子的水平并防止其毒性作用98,99。DHEA已被证明可以抑制有害的IL-6水平100。
炎症性疾病中DHEA的缺乏也意味着外周组织中各种性激素的缺乏,而DHEA是这些性激素的前体物。已知这些激素,包括雌激素和雄激素,对肌肉、骨骼和血管都有有益作用。然而,糖皮质激素的主流治疗会降低雄激素水平。因此,研究人员认为,患有慢性炎症性疾病的患者的激素替代不仅应包括糖皮质激素,还应包括DHEA100,101。
18. IBD与营养不良:
肠道炎症可能无法正常吸收营养。因此,IBD患者容易出现营养不良和维生素缺乏102-104。
19. IBD与高同型半胱氨酸:
许多研究表明,IBD患者的同型半胱氨酸水平更可能升高。对已发表研究的全面回顾发现,IBD患者患高同型半胱氨酸水平的风险是对照组的四倍多105。在一项研究中,超过55%的IBD患者的同型半胱氨酸水平升高106。IBD患者同型半胱氨酸升高的最大风险因素是叶酸水平降低107。维生素B12缺乏症也经常出现108。
IBD患者典型的同型半胱氨酸水平升高导致血栓和血管疾病的风险增加3倍109,110。这也有助于解释为什么IBD患者更有可能出现早期动脉粥样硬化111。
某些用于治疗IBD的药物,如甲氨蝶呤,是叶酸的抗代谢药物,这可能有助于解释为什么这么多患者缺乏叶酸。补充叶酸也可以减少甲氨蝶呤引起的不良反应112。
遗传学研究发现叶酸代谢的改变与IBD有关113。因此,IBD患者可能受益于补充5-甲基四氢叶酸(活性叶酸)。
有关管理同型半胱氨酸水平的更多相关内容,可参阅本网专文:高同型半胱氨酸 >>
20.IBD与骨质疏松症:
骨质疏松症是IBD的一种严重并发症,尽管其发病率高且具有潜在的破坏性影响,但尚未得到充分的认识114,115。骨质疏松症可能是由IBD本身引起的,也可能是糖皮质激素治疗的不良反应。来自对245名IBD患者的回顾性调查的数据表明,溃疡性结肠炎和克罗恩病患者骨折的患病率出乎意料地高,尤其是在病程长、活动期频繁、糖皮质激素摄入累积剂量高的患者中116,117。
此外,维生素D和维生素K水平低也与IBD患者骨质疏松症发生率较高有关118。IBD患者应定期进行骨密度测量,以预测骨折风险并确定预防和治疗阈值119。糖皮质激素也会增加骨质疏松症的风险,因为它们会影响钙和骨代谢。糖皮质激素抑制小肠对钙的吸收,增加肾脏对钙的排泄,并改变蛋白质代谢。服用糖皮质激素的克罗恩病患者与不服用的患者相比,骨折的风险更高120。有助于防止骨质流失的营养物质包括钙、镁、维生素D和维生素K。
更多相关内容,可参阅本网站专文:骨质疏松症 >>
21.IBD与血栓:
IBD患者形成血栓的风险增加,主要是静脉血栓栓塞121-123。这些凝块会破裂并滞留在肺部的血管中,可能导致死亡。此外,IBD患者使用糖皮质激素会增强凝血倾向121。传统药物通常依赖华法林或肝素来减轻IBD患者的血栓风险,但这些药物容易产生负面副作用,需要临床监测124。
维生素E、维生素D和白藜芦醇都可能有助于降低IBD患者凝血的风险125。了解更多血栓管理相关内容,可参阅本网站专文:血栓预防 >>
22. 叶酸与结肠癌风险:
溃疡性结肠炎患者患结肠癌的风险增加126。假设慢性炎症是导致溃疡性结肠炎癌症的原因。这得到了以下事实的支持:结肠癌癌症风险随着结肠炎持续时间的延长、结肠炎解剖范围的扩大以及其他炎症表现的伴随而增加127。叶酸缺乏和同型半胱氨酸水平升高与IBD中更大的结肠癌癌症风险有关128。
在一项涉及13项研究和725,000多名受试者数据的综合综述中,叶酸摄入量每增加100mcg/天,结肠癌癌症风险就会降低2%129。其他证据强调了叶酸在溃疡性结肠炎中预防结肠癌癌症的多种方法130。然而,数据是相互矛盾的,因为其他研究得出了不同的结论。例如,另一项综述发现,长期补充叶酸与结肠癌癌症风险增加有关131。
在IBD中经常观察到叶酸和维生素B12缺乏132。在饮食中补充维生素B12可以使身体更好地代谢叶酸,避免掩盖B12缺乏症。补充维生素B12很重要,尤其是对老年人和素食主义者,而且素食者饮食中摄入的B12很少。
更多内容可点击其个性化的综合干预方案如下:
以及参阅本网如下专文的相关内容:
医疗干预
大多数药物对治疗IBD的重点在于抗炎、免疫抑制,以减少肿胀和刺激,包括如下:
其他疗法
参考文献:
1. De Moreno de Leblanc A et al. Importance of IL-10 modulation by probiotic microorganisms in gastrointestinal inflammatory diseases. SRN Gastroenterol. 2011;2011:892971.
2. Lavasani S et al. A novel probiotic mixture exerts a therapeutic effect on experimental autoimmune encephalomyelitis mediated by IL-10 producing regulatory T cells. PLoS One. 2010 Feb 2;5(2):e9009.
3. Chin J. Intestinal microflora: negotiating health outcomes with the warring community within us. Asia Pac J Clin Nutr. 2004;13(suppl):S24-S25.
4. Fedorak RN et al. Probiotics and the management of inflammatory bowel disease. Inflamm Bowel Dis. 2004 May;10(3):286-99.
5. Furrie E et al. Systemic antibodies towards mucosal bacteria in ulcerative colitis and Crohn's disease differentially activate the innate immune response. Gut. 2004 Jan;53(1):91-8.
6. Sartor RB. Efficacy of probiotics for the management of inflammatory bowel disease. Gastroenterol Hepatol (N Y). 2011 Sep;7(9):606-8.
7. Rogler G. Prebiotics and probiotics in ulcerative colitis: where do we stand? Digestion. 2011;84(2):126-7.
8. Ishikawa H et al. Beneficial effects of probiotic bifidobacterium and galacto-oligosaccharide in patients with ulcerative colitis: a randomized controlled study. Digestion. 2011;84(2):128-33.
9. Fujimori S et al. High dose probiotic and prebiotic cotherapy for remission induction of active Crohn's disease. J Gastroenterol Hepatol. 2007;22(8):1199-204.
10. Karimi O et al. Probiotics (VSL#3) in arthralgia in patients with ulcerative colitis and Crohn's disease: A pilot study. Drugs Today (Barc) 2005;41(7):453-9.
11. Azcarate-Peril Ma et al. The intestinal microbiota, gastrointestinal environment and colorectal cancer: a putative role for probiotics in prevention of colorectal cancer? Am J Physiol Gastrointest Liver Physiol. 2011 Sep;301(3):G401-24.
12. Dinleyici Ec et al. Effectiveness and safety of Saccharomyces boulardii for acute infectious diarrhea. Expert Opin Biol Ther. 2012 Apr;12(4):395-410.
13. Thomas S et al. Anti-inflammatory effects of Saccharomyces boulardii mediated by myeloid dendritic cells from patients with Crohn's disease and ulcerative colitis. Am J Physiol Gastrointest Liver Physiol. 2011 Dec;301(6):G1083-92.
14. Garcia Vilela E et al. Influence of Saccharomyces boulardii on the intestinal permeability of patients with Crohn's disease in remission. Scand J Gastroenterol. 2008;43(7):842-8.
15. McFarland LV. Systematic review and meta-analysis of Saccharomyces boulardii in adult patients. World J Gastroenterol. 2010 May 14;16(18):2202-22.
16. Deckelbaum RJ et al. The omega-3 Fatty Acid nutritional landscape: health benefits and sources. J Nutr. 2012 Mar;142(3):587S-91S. Epub 2012 Feb 8.
17. Iwami D et al. Immunomodulatory effects of eicosapentaenoic acid through induction of regulatory T cells. Int Immunopharmacol. 2011 Mar;11(3):384-9.
18. Almallah YZ et al. Distal procto-colitis, natural cytotoxicity, and essential fatty acids. Am J Gastroenterol. 1998 May;93(5):804-9.
19. Hillier K et al. Incorporation of fatty acids from fish oil and olive oil into colonic mucosal lipids and effects upon eicosanoid synthesis in inflammatory bowel disease. Gut 1991;32(10):1151-5.
20. Steinhart AH et al. Nutrition in inflammatory bowel disease. Curr Opinion Gastroenterol. 1997;13(2):140-5.
21. Golias C et al. Physiology and pathophysiology of selectins, integrins, and IgSF cell adhesion molecules focusing on inflammation. A paradigm model on infectious endocarditis. ll Commun Adhes. 2011 Jun;18(3):19-32.
22. Ibrahim A et al. Dietary α-linolenic acid-rich formula reduces adhesion molecules in rats with experimental colitis. Nutrition. 2012 Jan 18.
23. Uchiyama K et al. N-3 polyunsaturated fatty acid diet therapy for patients with inflammatory bowel disease. Inflamm Bowel Dis. 2010 Oct;16(10):1696-707.
24. Stenson WF et al. Dietary supplementation with fish oil in ulcerative colitis. Ann Intern Med. 1992 Apr 15;116(8):609-14.
25. Aslan A et al. Fish oil fatty acid supplementation in active ulcerative colitis: a double-blind, placebo-controlled, crossover study. Am J Gastroenterol. 1992 Apr;87(4):432-7.
26. Wiese DM et al. The effects of an oral supplement enriched with fish oil, prebiotics, and antioxidants on nutrition status in Crohn's disease patients. Nutr Clin Pract. 2011 Aug;26(4):463-73.
27. Hawthorne AB et al. Treatment of ulcerative colitis with fish oil supplementation: a prospective 12 month randomised controlled trial. Gut 1992;33(7):922-8.
28. Belluzzi A et al. Effect of enteric-coated fish-oil preparation on relapses in Crohn's disease. N Engl J Med 1996;334(24):1557-60.
29. Simopoulos AP. Importance of the omega-6/omega-3 balance in health and disease: evolutionary aspects of diet. World Rev Nutr Diet. 2011;102:10-21.
30. Simopoulos AP. The importance of the ratio of omega-6/omega-3 essential fatty acids. Biomed Pharmacother. 2002 Oct;56(8):365-79.
31. Chambers ES et al. The impact of vitamin D on regulatory T cells. Curr Allergy Asthma Rep. 2011 Feb;11(1):29-36.
32. Ooi JH et al. Vitamin D regulation of immune function in the gut: why do T cells have vitamin D receptors? Mol Aspects Med. 2012 Feb;33(1):77-82.
33. Jahnsen J et al. Vitamin D status, parathyroid hormone and bone mineral density in patients with inflammatory bowel disease. Scand J Gastroenterol 2002;37(2):192-9.
34. Wang TT et al. Direct and indirect induction by 1,25-dihydroxyvitamin D3 of the NOD2/CARD15-defensin beta-2 innate immune pathway defective in Crohn disease. J Biol Chem 2010;285(4):2227-31.
35. Lim WC et al. Mechanisms of disease: vitamin D and inflammatory bowel disease. Nat Clin Pract Gastroenterol Hepatol 2005;2(7):308-15
36. Miheller P et al. Comparison of the effects of 1,25 dihydroxyvitamin D and 25 hydroxyvitamin D on bone pathology and disease activity in Crohn's disease patients. Inflamm Bowel Dis 2009;15:1656-1662.
37. Jorgensen SP et al. Clinical trial: Vitamin D3 treatment in Crohn's disease--A randomized double-blind placebo-controlled study. Aliment Pharmacol Ther 2010;32:377-83.
38. Abitbol V et al. Osteoporosis in inflammatory bowel disease: effect of calcium and vitamin D with or without fluoride. Aliment Pharmacol Ther. 2002 May;16(5):919-27.
39. Almeiner HA et al. Oxidative stress and inflammatory bowel disease. Front Biosci 2012;E4:1335-44.
40. Koutroubakis IE et al. Decreased total and corrected antioxidant capacity in patients with inflammatory bowel disease. Dig Dis Sci. 2004 Sep;49(9):1433-7.
41. Kruidenier L et al. Intestinal oxidative damage in inflammatory bowel disease: semi-quantification, localization, and association with mucosal antioxidants. J Pathol. 2003 Sep;201(1):28-36.
42. Trebble TM et al. Fish oil and antioxidants alter the composition and function of circulating mononuclear cells in Crohn disease. Am J Clin Nutr. 2004 Nov;80(5):1137-44.
43. Trebble TM et al. High-dose fish oil and antioxidants in Crohn's disease and the response of bone turnover: a randomised controlled trial. Br J Nutr. 2005 Aug;94(2):253-61.
44. Hengstermann S et al. Altered status of antioxidant vitamins and fatty acids in patients with inactive inflammatory bowel disease. Clin Nutr. 2008 Aug;27(4):571-8.
45. Babickova J et al. Pathological and therapeutic interactions between bacteriophages, microbes and the host in inflammatory bowel disease. World journal of gastroenterology: WJG. Oct 28 2015;21(40):11321-11330.
46. McCarville JL et al. Novel perspectives on therapeutic modulation of the gut microbiota. Therapeutic advances in gastroenterology.Jul 2016;9(4):580-593.
47. Clokie MRJ et al. Phages in nature. Bacteriophage.Jan-Feb 2011;1(1):31-45.
48. Belizario JE et al. Human microbiomes and their roles in dysbiosis, common diseases, and novel therapeutic approaches. Front Microbiol.2015;6:1050.
49. Norman JM et al. Disease-specific alterations in the enteric virome in inflammatory bowel disease. Cell.Jan 29 2015;160(3):447-460.
50. Wang W et al. Metagenomic Analysis of Microbiome in Colon Tissue from Subjects with Inflammatory Bowel Diseases Reveals Interplay of Viruses and Bacteria. Inflamm Bowel Dis.Jun 2015;21(6):1419-1427.
51. Loc-Carrillo C et al. Pros and cons of phage therapy. Bacteriophage.Mar-Apr 2011;1(2):111-114.
52. Bruttin A et al. Human volunteers receiving Escherichia coli phage T4 orally: a safety test of phage therapy. Antimicrobial agents and chemotherapy.Jul 2005;49(7):2874-2878.
53. Sarker SA et al. Oral T4-like phage cocktail application to healthy adult volunteers from Bangladesh. Virology.Dec 20 2012;434(2):222-232.
54. Taylor RA et al. Curcumin for inflammatory bowel disease: a review of human studies. Altern Med Rev. 2011 Jun;16(2):152-6.
55. Holt PR et al. Curcumin therapy in inflammatory bowel disease: a pilot study. Dig Dis Sci. 2005 Nov;50(11):2191-3.
56. Hanai H et al. Curcumin maintenance therapy for ulcerative colitis: Randomized, multicenter, double-blind, placebo-controlled trial. Clin Gastroenterol Hepatol 2006;4(12):1502-6.
57. Gerhardt H et al. Therapy of active Crohn disease with Boswellia serrata extract H 15. Z Gastroenterol 2001;39:11-7 [in German].
58. Gupta I et al. Effects of gum resin of Boswellia serrata in patients with chronic colitis. Planta Med 2001;67:391–5.
59. Gupta I et al. Effects of Boswellia serrata gum resin in patients with ulcerative colitis. Eur J Med Res 1997;2:37-43.
60. Holtmeier W et al. Randomized, placebo-controlled, double-blind trial of Boswellia serrata in maintaining remission of Crohn's disease: good safety profile but lack of efficacy. Inflamm Bowel Dis 2011;17(2):573-82.
61. Sengupta K et al. Cellular and molecular mechanisms of anti-inflammatory effect of Aflapin: a novel Boswellia serrata extract. Mol Cell Biochem. 2011 Aug;354(1-2):189-97. Epub 2011 Apr 11.
62. Vieira EL et al. Oral administration of sodium butyrate attenuates inflammation and mucosal lesion in experimental acute ulcerative colitis. J Nutr Biochem. 12 Jun 2011, 23(5):430-436
63. Segain JP et al. Butyrate inhibits inflammatory responses through NFkappaB inhibition: implications for Crohn's disease. Gut. 2000 Sep;47(3):397-403.
64. Assisi RF; GISDI Study Group. Combined butyric acid/mesalazine treatment in ulcerative colitis with mild-moderate activity. Results of a multicentre pilot study. Minerva Gastroenterol Dietol 2008;54(3):231-8.
65. Di Sabatino A et al. Oral butyrate for mildly to moderately active Crohn's disease. Aliment Pharmacol Ther 2005;22(9):789-94.
66. Langmead L et al. Randomized, double-blind, placebo-controlled trial of oral aloe vera gel for active ulcerative colitis. Aliment Pharmacol Ther. 2004a Apr 1;19(7):739-47.
67. Langmead L et al. Anti-inflammatory effects of aloe vera gel in human colorectal mucosa in vitro. Aliment Pharmacol Ther. 2004b Mar 1;19(5):521-7.
68. Omer B et al. Steroid-sparing effect of wormwood (Artemisia absinthium) in Crohn's disease: A double-blind placebo-controlled study. Phytomedicine 2007;14(2-3):87-95.
69. Krebs S et al. Wormwood (Artemisia absinthium) suppresses tumour necrosis factor alpha and accelerates healing in patients with Crohn's disease - A controlled clinical trial. Phytomedicine. 2010 Apr;17(5):305-9.
70. Geerling BJ et al. Comprehensive nutritional status in recently diagnosed patients with inflammatory bowel disease compared with population controls. Eur J Clin Nutr 2000a;54(6):514-21.
71. Hinks IJ et al. Reduced concentration of selenium in mild Crohn's disease. J Clin Pathol 1988;41:198-201.
72. Ojuawo A et al. The serum concentrations of zinc, copper and selenium in children with inflammatory bowel disease. Cent Afr J Med. 2002 Sep-Oct;48(9-10):116-9.
73. Geerling BJ et al. Nutritional supplementation with N-3 fatty acids and antioxidants in patients with Crohn's disease in remission: effects on antioxidant status and fatty acid profile. Inflamm Bowel Dis 2000b;6(2):77-84.
74. Abd-Allah AR et al. Pro-inflammatory and oxidative stress pathways which compromise sperm motility and survival may be altered by L-carnitine. Oxid Med Cell Longev. 2009 Apr-Jun;2(2):73-81.
75. Buyse J et al. Dietary L-carnitine supplementation enhances the lipopolysaccharide-induced acute phase protein response in broiler chickens. Vet Immunol Immunopathol. 2007 Jul 15;118(1-2):154-9.
76. Shakeri A et al. Effects of L-carnitine supplement on serum inflammatory cytokines, C-reactive protein, lipoprotein (a), and oxidative stress in hemodialysis patients with Lp (a) hyperlipoproteinemia. Hemodial Int. 2010 Oct;14(4):498-504.
77. Yuan Y et al. Protective effects of L-carnitine on intestinal ischemia/reperfusion injury in a rat model. J Clin Med Res. 2011 Apr 4;3(2):78-84
78. Mikhailova TL et al. Randomised clinical trial: the efficacy and safety of propionyl-L-carnitine therapy in patients with ulcerative colitis receiving stable oral treatment. Aliment Pharmacol Ther. 2011 Nov;34(9):1088-97.
79. Den Hond E et al. Effect of long-term oral glutamine supplements on small intestinal permeability in patients with Crohn's disease. JPEN J Parenter Enteral Nutr 1999;23(1):7-11.
80. Kruschewski M et al. [Protective effect of glutamine on microcirculation of the intestine in experimental colitis]. Langenbecks Arch Chir Suppl Kongressbd. 1998;115(Suppl I):229-31.
81. Sido B et al. Low intestinal glutamine level and low glutaminase activity in Crohn's disease: a rational for glutamine supplementation? Dig Dis Sci 2006;51(12):2170-9.
82. Benjamin J et al. Glutamine and Whey Protein Improve Intestinal Permeability and Morphology in Patients with Crohn's Disease: A Randomized Controlled Trial. Dig Dis Sci. 2011 Oct 26.
83. Ockenga J et al. Glutamine-enriched total parenteral nutrition in patients with inflammatory bowel disease. Eur J Clin Nutr. 2005 Nov;59(11):1302-9.
84. Krasinski SD, Russell RM et al. The prevalence of vitamin K deficiency in chronic gastrointestinal disorders. Am J Clin Nutr 1985;41(3):639-43.
85. Nakajima S et al. Association of vitamin K deficiency with bone metabolism and clinical disease activity in inflammatory bowel disease. Nutrition 2011;27(10):1023-8.
86. Duggan P et al. Vitamin K status in patients with Crohn's disease and relationship to bone turnover. Am J Gastroenterol 2004;99(11):2178-85.
87. Hou JK et al. Dietary intake and risk of developing inflammatory bowel disease: a systematic review of the literature. Am J Gastroenterol. 2011 Apr;106(4):563-73.
88. Sakamoto N et al. Dietary risk factors for inflammatory bowel disease: a multicenter case-control study in Japan. Inflamm Bowel Dis. 2005 Feb;11(2):154-63.
89. Heaton KW et al. Treatment of Crohn's disease with an unrefined-carbohydrate, fiber-rich diet. Br Med J 1979;ii:764-6.
90. Hanai H, Kanauchi O, et al. Germinated barley foodstuff prolongs remission in patients with ulcerative colitis. Int J Mol Med 2004;13(5):643-7.
91. Bubenik GA. Gastrointestinal melatonin: localization, function, and clinical relevance. Dig Dis Sci 2002;47(10):2336-48.
92. Johe PD et al. The in vivo effect of melatonin on cellular activation processes in human blood during strenuous physical exercise. J Pineal Res 2005;39:324–330.
93. Terry PD et al. Melatonin and ulcerative colitis: evidence, biological mechanisms, and future research. Inflamm Bowel Dis 2009;15:134-40.
94. Boznanska P et al. 24-hour urinary 6-hydroxymelatonin sulfate excretion in patients with ulcerative colitis. Pol Merkur Lekarski 2007;22(131):369-72 [in Polish].
95. Chojnacki C et al. Evaluation of melatonin effectiveness in the adjuvant treatment of ulcerative colitis. J Physiol Pharmacol 2011;62(3):327-34.
96. Rakhimova OIu. Use of melatonin in combined treatment for inflammatory bowel diseases. Ter Arkh 2010;82(12):64-8 [in Russian].
97. Maldonado MD et al. Melatonin usage in ulcerative colitis: a case report. J Pineal Res 2008;45(3):339-40.
98. Haden ST et al. Effects of age on serum dehydroepiandrosterone sulfate, IGF-I, and IL-6 levels in women. Calcif Tissue Int. 2000 Jun;66(6):414-8.
99. Head KA et al. Inflammatory bowel disease Part 1: ulcerative colitis--pathophysiology and conventional and alternative treatment options. Altern Med Rev. 2003 Aug;8(3):247-83.
100. Andus T et al. Patients with refractory Crohn's disease or ulcerative colitis respond to dehydroepiandrosterone: a pilot study. Aliment Pharmacol Ther. 2003 Feb;17(3):409-14.
101. Straub RH et al. Replacement therapy with DHEA plus corticosteroids in patients with chronic inflammatory diseases: substitutes of adrenal and sex hormones. Z Rheumatol. 2000;59(suppl 2):II/108-18.
102. Alastair F et al. Nutrition in inflammatory bowel disease. JPEN J Parenter Enteral Nutr. 2011 Sep;35(5):571-80.
103. Mortimore M et al. A role for B₁₂ in inflammatory bowel disease patients with suppurative dermatoses? An experience with high dose vitamin B₁₂ therapy. J Crohns Colitis. 2010 Oct;4(4):466-70.
104. Campos FG et al. Pharmacological nutrition in inflammatory bowel diseases. Nutr Hosp. 2003 Mar;18(2):57-64.
105. Oussalah A et al. ta-analysis: hyperhomocysteinaemia in inflammatory bowel diseases. Aliment Pharmacol Ther. 2011 Nov;34(10):1173-84.
106. Roblin X et al. Factors associated with hyperhomocysteinemia in inflammatory bowel disease: prospective study in 81 patients [in French]. Rev Med Interne. 2006 Feb;27(2):106-10.
107. Zezos P et al. Hyperhomocysteinemia in ulcerative colitis is related to folate levels. World J Gastroenterol. 2005 Oct 14;11(38):6038-42.
108. Mahmood A et al. Prevalence of hyperhomocysteinaemia, activated protein C resistance and prothrombin gene mutation in inflammatory bowel disease. Eur J Gastroenterol Hepatol 2005;17:739-44.
109. Fernandez-Miranda C et al. Hyperhomocysteinemia and methylenetetrahydrofolate reductase 677C-->T and 1298A-->C mutations in patients with inflammatory bowel disease. Rev Esp Enferm Dig. 2005 Jul;97(7):497-504.
110. Srirajaskanthan R et al. Venous thrombosis in inflammatory bowel disease. Eur J Gastroenterol Hepatol. 2005 Jul;17(7):697-700.
111. Papa A et al. Increased carotid intima-media thickness in patients with inflammatory bowel disease. Aliment Pharmacol Ther. 2005 Nov 1;22(9):839-46.
112. Patel V et al. Methotrexate for maintenance of remission in Crohn's disease. Cochrane Database Syst Rev 2009 Oct 7;(4):CD006884.
113. Zintzaras E. Genetic variants of homocysteine/folate metabolism pathway and risk of inflammatory bowel disease: a synopsis and meta-analysis of genetic association studies. Biomarkers. 2010 Feb;15(1):69-79.
114. Etzel JP et al. Assessment and management of low bone density in inflammatory bowel disease and performance of professional society guidelines. Inflamm Bowel Dis. 2011 Jan 13.
115. Harpavat M et al. Metabolic bone disease in inflammatory bowel disease. J Clin Gastroenterol. 2004 Mar;38(3):218-24.
116. Miheller P et al. Clinical relevance of changes in bone metabolism in inflammatory bowel disease. World J Gastroenterol. 2010 Nov 28;16(44):5536-42.
117. Agrawal M et al. Bone, inflammation, and inflammatory bowel disease. Curr Osteoporos Rep. 2011 Dec;9(4):251-7.
118. Kuwabara A et al. High prevalence of vitamin K and D deficiency and decreased BMD in inflammatory bowel disease. Osteoporos Int 2009;20(6):935-42.
119. Rogler G et al. Extraintestinal manifestations of inflammatory bowel disease [in German]. Med Klin (Munich). 2004 Mar 15;99(3):123-30.
120. Bernstein CN et al. The association between corticosteroid use and development of fractures among IBD patients in a population-based database. Am J Gastroenterol 2003;98(8):1797-801.
121. Kappelman MD et al. Thromboembolic risk among Danish children and adults with inflammatory bowel diseases: a population-based nationwide study. Gut 2011;60:937–43.
122. Solem CA et al. Venous thromboembolism in inflammatory bowel disease. Am J Gastroenterol 2004;99(1):97-101.
123. Sonoda K et al. Evaluation of venous thromboembolism and coagulation-fibrinolysis markers in Japanese patients with inflammatory bowel disease. J Gastroenterol 2004;39(10):948-54.
124. Koutroubakis IE. Therapy insight: Vascular complications in patients with inflammatory bowel disease. Nat Clin Pract Gastroenterol Hepatol 2005;2(6):266-72.
125. Phang M et al. Diet and thrombosis risk: nutrients for prevention of thrombotic disease. Semin Thromb Hemost 2011;37(3):199-208.
126. Mitamura T et al. The more an ulcerative colitis is repeated, the more the risk of colorectal carcinogenesis is increased in mice. Anticancer Res. 2002 Nov-Dec;22(6C):3955-61.
127. Itzkowitz SH et al. Inflammation and cancer IV. Colorectal cancer in inflammatory bowel disease: the role of inflammation. Am J Physiol Gastrointest Liver Physiol. 2004 Jul;287(1):G7-G17.
128. Phelip JM et al. Association of hyperhomocysteinemia and folate deficiency with colon tumors in patients with inflammatory bowel disease. Inflamm Bowel Dis. 2008 Feb;14(2):242-8.
129. Kim DH et al. Pooled analyses of 13 prospective cohort studies on folate intake and colon cancer. Cancer Causes Control. 2010 Nov;21(11):1919-30. Epub 2010 Sep 5.
130. Biasco G et al. Folate and prevention of colorectal cancer in ulcerative colitis. Eur J Cancer Prev. 2005 Aug;14(4):395-8.
131. Fife J et al. Folic acid supplementation and colorectal cancer risk: a meta-analysis. Colorectal Dis. 2011 Feb;13(2):132-7.
132. Yakut M et al. Serum vitamin B12 and folate status in patients with inflammatory bowel diseases. Eur J Intern Med. 2010 Aug;21(4):320-3.
美国国立公众健康网
www.medlineplus.gov
美国胃肠病协会
http://www.gastro.org
美国家庭医生学会
http://www.familydoctor.org
美国国立糖尿病、消化和肾病研究所
http://www.niddk.nih.gov
加拿大胃肠病协会
https://www.cag-acg.org
加拿大卫生部
http://www.hc-sc.gc.ca
免责声明和安全信息
英文名称:Inflammatory Bowel Disease
定义
炎症性肠病(IBD)表现为肠道肿胀和刺激症状,是由于肠腔和肠内壁的免疫失衡造成的。IBD是终生性疾病,并可导致严重腹泻、腹痛、疲劳和体重下降。IBD可逐渐导致患者衰弱,甚至可能危及生命的并发症。抑制炎症是其治疗的首要目标,然而,主要的抗炎药物如糖皮质激素有很多副作用,大大限制了其长期治疗和疗效。IBD主要分为有2种类型:
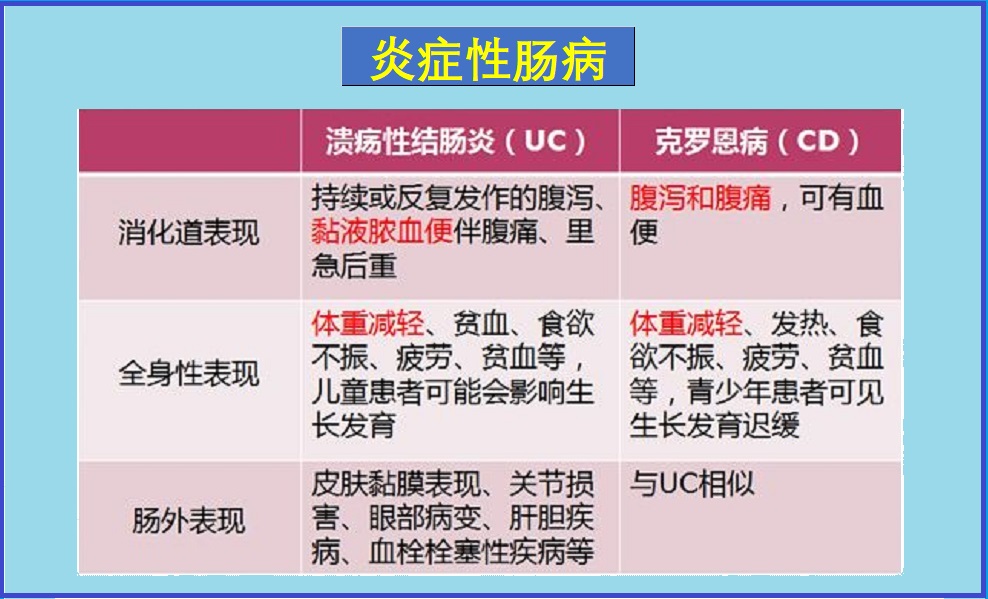
- 溃疡性结肠炎:在大肠(结肠)、直肠造成持久性的炎症和溃疡。
- 克罗恩病:主要在小肠最后一部分(回肠)但却可以出现在消化道内层中,及常扩散到受影响的相关组织。
病因
炎症性肠病的确切病因尚不清楚,据认为可能是以下原因造成:- 遗传基因,家族IBD病史
- 对损伤结肠和直肠的病毒或细菌的反应
- 受损的免疫系统,或影响免疫系统的感染
风险因素
大多数患IBD的人在30岁之前就可被诊断出来。下列因素可增加罹患IBD几率:
- 家庭成员有IBD
- 免疫系统问题
- 经常使用非甾体类抗炎药NSAIDs
- 吸烟
症状
症状可能是经常性的,或仅在发作时出现。症状也取决于IBD的类型。常见症状包括:
- 腹部疼痛和抽筋
- 腹泻
- 疲劳乏力
- 缺乏食欲、体重下降
- 肠道出血
- 溃疡肠道
- 直肠炎症
- 腹胀或满腹感觉
- 积气
- 腹泻及带血
- 腹部有声音
- 恶心和呕吐
- 关节疼痛
并发症
一般并发症可能包括如下:- 结肠癌:患IBD会增加患结肠癌的风险。
- 皮肤、眼睛和关节问题:IBD发作期间可能会出现某些并发症,包括关节炎、眼部炎症(葡萄膜炎)和皮肤损伤。
- 药物副作用:皮质类固醇可能与骨质疏松症、高血压和其他疾病风险有关。某些治疗IBD的药物与一些癌症有关。
- 原发性硬化性胆管炎:炎症引起胆管疤痕,使它们变窄并逐渐引起肝损伤。
- 血栓:IBD增加静脉和动脉血栓的风险。
此外,溃疡性结肠炎和克罗恩病由可能发展出不同的并发症,详细可参考本网有关专文。
疗法
IBD不能治愈,但一些治疗可帮助控制症状。选项可包括如下:
调整饮食与生活方式
- 低脂肪饮食。
- 低纤维和乳制品,在医生或专业营养师指导下。
- 避免刺激性食物,如咖啡因、酒精和辛辣食物等
- 学会各种放松、减轻压力技巧。
- 适当的运动锻炼。
营养与草本综合干预
以下是基于循证医学和循证营养学有关文献综合的结果。
有助于防控炎症性肠病的营养和草本补充剂,主要包括如下:
1.益生菌:
消化道内微生物种群的变化能够局部和系统地改变免疫细胞的功能。一项研究描述了一种新型益生菌,其可以直接产生白细胞介素-10(IL-10),这是一种促进免疫耐受的抗炎细胞因子1-3。此外,摄入益生菌可以通过各种机制削弱致病菌的作用,包括竞争上皮受体结合和增强肠道屏障功能1,4,5。一些益生菌还产生丁酸,一种对结肠壁内细胞健康很重要的短链脂肪酸(见下文)6。
益生菌在IBD人群中使用的临床试验表明了有益的效果。试验的持续时间和使用的生物体各不相同,但有几次显示积极的结果7。2011年一项使用益生菌(短双歧杆菌)和益生元(低聚半乳糖)的试验表明,溃疡性结肠炎患者的临床状况显著改善8。克罗恩病的临床试验表明,每天供应500亿个或更多的益生菌生物体补充剂(如ACTIAL益生菌VSL#3)改善了肠道健康9,10。在一项试验中,两名受试者的缓解效果非常好,他们能够停止糖皮质激素药物治疗9。其他研究表明,益生菌可能抑制结直肠癌癌症发展的可能性,这是IBD患者的主要关注问题11。
另一种在IBD中显示出前景的生物是布拉氏酵母。几项试验已经证明布拉氏酵母菌对改善感染性腹泻和其他胃肠道问题的疗效12。此外,与IBD特别相关的布拉氏酵母菌似乎可以调节肠上皮的炎症反应,降低TNF-α和IL-613。同一项研究表明,布拉氏酵母促进IBD患者细胞样本中的肠道组织修复和免疫耐受。在一项随机、安慰剂对照的临床试验中,布拉氏酵母菌在常规治疗中添加后,降低了克罗恩病患者的肠道通透性14。在各种病理状态下,补充布拉氏酵母菌通常是安全有效的15。
2. 欧米伽3脂肪酸:
两种最主要的ω-3脂肪酸即二十碳五烯酸(EPA)和二十二碳六烯酸(DHA),存在于冷水鱼中16。Omega-3脂肪酸是强大的免疫调节剂,可减少循环炎症细胞因子并降低自然杀伤细胞的细胞毒性17-20。此外,在一项动物研究中,α-亚麻酸(一种植物衍生的ω-3脂肪酸)抑制了粘附分子的表达,粘附分子在炎症、免疫反应和细胞内信号事件中很重要21,22。
在临床试验中,补充鱼油可以改善克罗恩病和溃疡性结肠炎患者的脂肪酸状况,并与较低水平的炎症介质有关23-25。这些变化与疾病发作的缓解有一定的相关性26,27。鱼油还可以减少缓解所需的糖皮质激素药物的剂量27。在一项针对克罗恩病患者的研究中发现,肠溶鱼油制剂有助于降低复发率28。
现代饮食导致多数人血液中ω-6和ω-3的比例不健康,这种失衡与炎症性疾病密切相关29。一般建议将ω-6与ω-3的比例保持在4:1以下,以获得最佳健康30;这对于IBD患者可能特别重要。
3. 维生素D:
维生素D是另一种强大的免疫调节剂。实验模型表明,T细胞表达维生素D受体,缺乏维生素D信号会导致T细胞产生更高水平的炎症细胞因子。此外,维生素D是Treg细胞亚群发育所必需的,Treg细胞在抑制肠道炎症方面具有重要作用31,32。IBD患者的维生素D水平通常不足,如血清25-羟基维生素D水平较低所示33。许多其他证据也将维生素D水平低与IBD联系起来34,35。
在37名克罗恩病缓解期患者中,服用25-羟基维生素D3或骨化三醇(活性维生素D3,处方药)降低了炎症指标,改善了骨骼健康36。在一项涉及94名克罗恩病缓解期患者的双盲试验中,与安慰剂相比,每天服用1200IU维生素D3显示出复发率较低的趋势(从29%降至13%[P=0.06])37。此外,骨丢失是IBD患者的主要担忧,这种疾病以及用于治疗它的糖皮质激素都会导致骨骼健康状况不佳。补充维生素D已被证明可以在克罗恩病中保持骨密度38。
通常建议将血清25-羟基维生素D水平维持在50-80ng/mL的范围内。补充维生素D的人应定期进行25-羟基维生素D血液测试,以确保其保持在最佳范围内。
4. 维生素抗氧化剂:
抗氧化剂。正常消化会产生大量活性氧和氮物质(也称为自由基),肠粘膜对其保持广泛的抗氧化剂防御系统。然而,当出现过度氧化应激时,粘膜屏障会持续受损并渗漏,从而为炎症奠定基础39,40。此外,炎症本身会产生大量的反应性物种,破坏性的循环可能会持续下去。在患有IBD的患者中,肠道中存在高水平的活性氧,这会导致疾病造成的损害39。
在一项研究中,IBD患者的抗氧化能力明显低于非IBD患者41。一些研究表明,维生素A、维生素C、维生素E和硒的抗氧化剂组合与鱼油结合可以降低克罗恩病的某些炎症标志物42,43。此外,IBD患者血液中的类胡萝卜素和维生素C水平明显较低44。
5. 噬菌体:
噬菌体(Bacteriophages)是以细菌为目标的病毒。它们是地球上最丰富的生物,人类消化道估计含有1015个噬菌体45-47。肠道噬菌体似乎在肠道微生物群落的生态学中发挥着重要作用,包括将病毒遗传物质注入到特定细菌中,在某些情况下导致其快速死亡46-48。尽管噬菌体数量丰富,但直到最近,它们还相对较少受到研究关注46,48。新出现的证据表明噬菌体在治疗炎症性肠病方面具有治疗潜力46,45。
噬菌体似乎可以调节免疫活性,并可能影响肠道内壁的炎症。在患有炎症性肠病的个体中已经注意到噬菌体群体的疾病特异性模式,并且与在健康个体中观察到的噬菌体群体有显著差异49,50;此外,在一项研究中发现,某些噬菌体的丰度与某些类型细菌水平的降低有关49。
噬菌体疗法可能比抗生素治疗更安全,部分原因是它对正常肠道菌群的破坏最小51。在一项安全性研究中,口服补充针对肠道细菌大肠杆菌的噬菌体导致健康志愿者的粪便中可检测到这些噬菌体的存在,但在补充结束后的一周内不再检测到它们的存在。非致病性大肠杆菌的数量保持不变,没有发现不良副作用52。同样,在一项针对健康成年人的研究中,服用九种大肠杆菌靶向噬菌体的混合物,在噬菌体给药后立即收集的粪便样本中未检测到大肠杆菌,在血液、肝脏和肾脏测试中也未报告或检测到副作用53。
6.姜黄素:
姜黄素作为抗炎剂在各种环境中的功效已得到充分证明。其多重作用中最突出的是抑制核因子κB(NF-kB)信号传导。NF-kB是一种信号蛋白,它驱动无数炎症细胞因子的产生,包括白细胞介素1b(IL-1b)和白细胞介素-6(IL-6)。由于NF-kB和相关细胞因子是IBD病理学的核心,姜黄素已被研究作为一种干预措施54。
在一项研究中,姜黄素有助于减轻一小群患者的克罗恩病和溃疡性结肠炎症状,其中许多患者能够停用氨基水杨酸盐和/或糖皮质激素54,55。在一组82名溃疡性结肠炎患者中,与安慰剂加氨基水杨酸盐相比,姜黄素联合氨基水杨酸盐降低了急性发作的复发和症状严重程度。姜黄素组在治疗的6个月内复发率为4.6%,而对照组的复发率超过20%56。
7.乳香:
乳香树脂含有一种强效抗炎化合物即乙酰基-11-keto-β-boswellic acid(AKBA),简称乳香酸。一项双盲临床试验发现,在改善克罗恩病症状方面,乳香与美沙拉秦一样有效,副作用要少得多57。一项试验还发现,在30名患者中,乳香与柳氮磺胺吡啶一样有效,可以诱导溃疡性结肠炎的缓解58。这证实了早期关于乳香治疗溃疡性结肠炎患者的疗效报告59。
然而,另一项涉及108名克罗恩病患者的双盲试验并未发现乳香在维持病情缓解方面优于安慰剂60。一种名为AprèsFlex™的改良提取物(或Aflapin®),它将AKBA与其他非挥发性的乳香油结合在一起,与标准化为相同百分比的AKBA的其他制剂相比,在较低浓度下显示出改善的抗炎活性61。
8.丁酸:
丁酸(或丁酸盐)是一种短链脂肪酸,由肠道中的纤维被某些细菌代谢时产生。实验模型表明,口服丁酸盐可改善溃疡性结肠炎的炎症62。丁酸可能发挥作用的一种机制是抑制促炎细胞信号传导成分核因子κB(NF-kB)的激活63。在临床试验中,口服丁酸盐对克罗恩病和溃疡性结肠炎都有缓解作用64,65。在一项试验中,近70%的克罗恩病受试者对每天4g丁酸肠溶片的剂量有反应,持续8周。在这些应答者中,53%的应答者病情缓解,他们的NF-kB和另一种炎症因子IL-1b水平显著下降65。
9.芦荟(凝胶):
芦荟叶内部的粘液凝胶多年来一直被用于治疗溃疡性结肠炎。一项双盲随机试验发现,每天两次3盎司(约85g)的芦荟凝胶比安慰剂更好地结束了溃疡性结肠炎患者的急性发作,没有不良反应66。芦荟凝胶的免疫调节、肠道愈合和抗炎特性都可能在其功效中发挥作用67。
10.艾蒿:
艾蒿(Wormwood,又称艾草或苦艾)是一种原产于地中海地区的苦味草本植物,其标准化提取物已在克罗恩病患者身上进行了研究。与安慰剂相比,它在逐渐减少药物治疗的患者中更有效地维持病情缓解68。这可能是因为艾草可以阻断TNF-α,一种强效的促炎细胞因子69。
11. 硒:
硒是一种微量元素,对许多硒依赖性酶的功能至关重要。硒缺乏在IBD患者中很常见70-72。补充有助于缓解这一问题,这既基于血清硒的增加,也基于谷胱甘肽过氧化物酶功能的改善73。
12. L-肉碱:
L肉碱是正常细胞代谢所必需的,而肉碱水平不足尤其会影响需要大量能量的细胞,如免疫系统细胞。几项实验表明,肉碱调节炎症介质的产生,并且肉碱水平不足与炎症细胞因子的大量产生有关74,75。事实上,在一项涉及36名透析患者的临床试验中,每天补充1g L-肉碱可使CRP水平降低29%,IL-6水平降低61%76。
就肠道而言,在动物模型中,L-肉碱显著减弱了肠道组织缺氧和恢复的炎症反应77。在一项涉及121名溃疡性结肠炎受试者的随机安慰剂对照试验中,每天1或2g的丙酰-L-肉碱与安慰剂相比,在常规治疗中的缓解率更高78。在每天服用1g L肉碱组中,病情缓解率为55%,而安慰剂组仅为35%。
13.谷氨酰胺:
这是一种条件必需氨基酸,也是肠细胞(肠道吸收细胞)的主要燃料。口服谷氨酰胺补充可以稳定肠道通透性和粘膜完整性79。一项研究表明,谷氨酰胺有助于改善结肠炎动物结肠发炎部分的毛细血管血流量80。此外,中度至重度克罗恩病患者的谷氨酰胺水平较低81。
在一项随机临床试验中,每天0.5g/kg体重的谷氨酰胺剂量持续2个月,可降低克罗恩病患者的肠道通透性并改善其形态82。然而,补充谷氨酰胺的临床益处可能仅限于缓解期,因为另一项试验发现,在疾病发作期间补充谷氨酰胺并不能改善肠道通透性83。
14. 维生素K:
IBD患者经常缺乏维生素K。一项研究表明,31%的溃疡性结肠炎或克罗恩病患者缺乏维生素K84。在一项研究中,维生素K活性低与克罗恩病活性高有关85。IBD患者的维生素K缺乏也与骨密度降低有关85,86。
15. 膳食纤维:
摄入更多的膳食纤维与克罗恩病的发病率较低有关87,而摄入更多的糖与风险增加有关88。低精制糖和高膳食纤维的饮食已被证明对克罗恩病的病程有良好的影响,并且与正常饮食相比不会导致肠梗阻89。
肠道细菌发酵膳食纤维是短链脂肪酸(如丁酸)的主要来源,各种研究表明,植物纤维有助于预防溃疡性结肠炎发作90。
16. 褪黑素:
虽然褪黑素是一种有助于同步睡眠-觉醒周期的激素,但它在消化道中的产生量远大于大脑91。褪黑素降低TNF-α水平92。许多体外和动物研究表明,褪黑素可以减轻IBD的炎症93。IBD患者的褪黑激素合成增加,较高的水平与较低的症状有关,这表明这是身体减少过度炎症的努力的一部分94。
在一项针对60名接受美沙拉秦治疗的溃疡性结肠炎患者的双盲试验中,一半患者随机服用褪黑素,一半患者服用安慰剂,为期一年95。安慰剂组的炎症和临床症状有所上升,而褪黑素组仍处于缓解状态。这证实了早期一项未经控制的研究,该研究表明褪黑素对克罗恩病和溃疡性结肠炎患者有帮助96。尽管如此,还是要谨慎——至少有一项案例研究发表,其中褪黑素导致溃疡性结肠炎发作,而对糖皮质激素没有反应97。
17. 脱氢表雄酮(DHEA):
DHEA在预防慢性炎症和维持健康免疫功能方面发挥着重要作用。已发表的研究将低水平的DHEA与慢性炎症联系起来,并且DHEA已被证明可以抑制促炎细胞因子的水平并防止其毒性作用98,99。DHEA已被证明可以抑制有害的IL-6水平100。
炎症性疾病中DHEA的缺乏也意味着外周组织中各种性激素的缺乏,而DHEA是这些性激素的前体物。已知这些激素,包括雌激素和雄激素,对肌肉、骨骼和血管都有有益作用。然而,糖皮质激素的主流治疗会降低雄激素水平。因此,研究人员认为,患有慢性炎症性疾病的患者的激素替代不仅应包括糖皮质激素,还应包括DHEA100,101。
18. IBD与营养不良:
肠道炎症可能无法正常吸收营养。因此,IBD患者容易出现营养不良和维生素缺乏102-104。
19. IBD与高同型半胱氨酸:
许多研究表明,IBD患者的同型半胱氨酸水平更可能升高。对已发表研究的全面回顾发现,IBD患者患高同型半胱氨酸水平的风险是对照组的四倍多105。在一项研究中,超过55%的IBD患者的同型半胱氨酸水平升高106。IBD患者同型半胱氨酸升高的最大风险因素是叶酸水平降低107。维生素B12缺乏症也经常出现108。
IBD患者典型的同型半胱氨酸水平升高导致血栓和血管疾病的风险增加3倍109,110。这也有助于解释为什么IBD患者更有可能出现早期动脉粥样硬化111。
某些用于治疗IBD的药物,如甲氨蝶呤,是叶酸的抗代谢药物,这可能有助于解释为什么这么多患者缺乏叶酸。补充叶酸也可以减少甲氨蝶呤引起的不良反应112。
遗传学研究发现叶酸代谢的改变与IBD有关113。因此,IBD患者可能受益于补充5-甲基四氢叶酸(活性叶酸)。
有关管理同型半胱氨酸水平的更多相关内容,可参阅本网专文:高同型半胱氨酸 >>
20.IBD与骨质疏松症:
骨质疏松症是IBD的一种严重并发症,尽管其发病率高且具有潜在的破坏性影响,但尚未得到充分的认识114,115。骨质疏松症可能是由IBD本身引起的,也可能是糖皮质激素治疗的不良反应。来自对245名IBD患者的回顾性调查的数据表明,溃疡性结肠炎和克罗恩病患者骨折的患病率出乎意料地高,尤其是在病程长、活动期频繁、糖皮质激素摄入累积剂量高的患者中116,117。
此外,维生素D和维生素K水平低也与IBD患者骨质疏松症发生率较高有关118。IBD患者应定期进行骨密度测量,以预测骨折风险并确定预防和治疗阈值119。糖皮质激素也会增加骨质疏松症的风险,因为它们会影响钙和骨代谢。糖皮质激素抑制小肠对钙的吸收,增加肾脏对钙的排泄,并改变蛋白质代谢。服用糖皮质激素的克罗恩病患者与不服用的患者相比,骨折的风险更高120。有助于防止骨质流失的营养物质包括钙、镁、维生素D和维生素K。
更多相关内容,可参阅本网站专文:骨质疏松症 >>
21.IBD与血栓:
IBD患者形成血栓的风险增加,主要是静脉血栓栓塞121-123。这些凝块会破裂并滞留在肺部的血管中,可能导致死亡。此外,IBD患者使用糖皮质激素会增强凝血倾向121。传统药物通常依赖华法林或肝素来减轻IBD患者的血栓风险,但这些药物容易产生负面副作用,需要临床监测124。
维生素E、维生素D和白藜芦醇都可能有助于降低IBD患者凝血的风险125。了解更多血栓管理相关内容,可参阅本网站专文:血栓预防 >>
22. 叶酸与结肠癌风险:
溃疡性结肠炎患者患结肠癌的风险增加126。假设慢性炎症是导致溃疡性结肠炎癌症的原因。这得到了以下事实的支持:结肠癌癌症风险随着结肠炎持续时间的延长、结肠炎解剖范围的扩大以及其他炎症表现的伴随而增加127。叶酸缺乏和同型半胱氨酸水平升高与IBD中更大的结肠癌癌症风险有关128。
在一项涉及13项研究和725,000多名受试者数据的综合综述中,叶酸摄入量每增加100mcg/天,结肠癌癌症风险就会降低2%129。其他证据强调了叶酸在溃疡性结肠炎中预防结肠癌癌症的多种方法130。然而,数据是相互矛盾的,因为其他研究得出了不同的结论。例如,另一项综述发现,长期补充叶酸与结肠癌癌症风险增加有关131。
在IBD中经常观察到叶酸和维生素B12缺乏132。在饮食中补充维生素B12可以使身体更好地代谢叶酸,避免掩盖B12缺乏症。补充维生素B12很重要,尤其是对老年人和素食主义者,而且素食者饮食中摄入的B12很少。
更多内容可点击其个性化的综合干预方案如下:
以及参阅本网如下专文的相关内容:
医疗干预
大多数药物对治疗IBD的重点在于抗炎、免疫抑制,以减少肿胀和刺激,包括如下:
- 抗炎药物
- 皮质类固醇
- 免疫抑制剂
- 抗生素
- 抗腹泻药物
- 泻药
- 止痛药
其他疗法
- 贫血与补铁:慢性肠道出血可致贫血和缺铁,需要及时补充。
- 骨松症与补充维生素D、钙:慢性肠炎导致吸收障碍,皮质类固醇药治疗也可引起骨松症。
- 特殊饮食营养支持:肠内营养和肠外营养支持,以支持肠道休息、减少炎症发作。
- 低残留饮食:如果肠道狭窄,可减少未消化的食物卡在肠狭窄部分而导致堵塞的可能性。
预防
因为病因不明确,还没有预防IBD的指南。参考文献:
1. De Moreno de Leblanc A et al. Importance of IL-10 modulation by probiotic microorganisms in gastrointestinal inflammatory diseases. SRN Gastroenterol. 2011;2011:892971.
2. Lavasani S et al. A novel probiotic mixture exerts a therapeutic effect on experimental autoimmune encephalomyelitis mediated by IL-10 producing regulatory T cells. PLoS One. 2010 Feb 2;5(2):e9009.
3. Chin J. Intestinal microflora: negotiating health outcomes with the warring community within us. Asia Pac J Clin Nutr. 2004;13(suppl):S24-S25.
4. Fedorak RN et al. Probiotics and the management of inflammatory bowel disease. Inflamm Bowel Dis. 2004 May;10(3):286-99.
5. Furrie E et al. Systemic antibodies towards mucosal bacteria in ulcerative colitis and Crohn's disease differentially activate the innate immune response. Gut. 2004 Jan;53(1):91-8.
6. Sartor RB. Efficacy of probiotics for the management of inflammatory bowel disease. Gastroenterol Hepatol (N Y). 2011 Sep;7(9):606-8.
7. Rogler G. Prebiotics and probiotics in ulcerative colitis: where do we stand? Digestion. 2011;84(2):126-7.
8. Ishikawa H et al. Beneficial effects of probiotic bifidobacterium and galacto-oligosaccharide in patients with ulcerative colitis: a randomized controlled study. Digestion. 2011;84(2):128-33.
9. Fujimori S et al. High dose probiotic and prebiotic cotherapy for remission induction of active Crohn's disease. J Gastroenterol Hepatol. 2007;22(8):1199-204.
10. Karimi O et al. Probiotics (VSL#3) in arthralgia in patients with ulcerative colitis and Crohn's disease: A pilot study. Drugs Today (Barc) 2005;41(7):453-9.
11. Azcarate-Peril Ma et al. The intestinal microbiota, gastrointestinal environment and colorectal cancer: a putative role for probiotics in prevention of colorectal cancer? Am J Physiol Gastrointest Liver Physiol. 2011 Sep;301(3):G401-24.
12. Dinleyici Ec et al. Effectiveness and safety of Saccharomyces boulardii for acute infectious diarrhea. Expert Opin Biol Ther. 2012 Apr;12(4):395-410.
13. Thomas S et al. Anti-inflammatory effects of Saccharomyces boulardii mediated by myeloid dendritic cells from patients with Crohn's disease and ulcerative colitis. Am J Physiol Gastrointest Liver Physiol. 2011 Dec;301(6):G1083-92.
14. Garcia Vilela E et al. Influence of Saccharomyces boulardii on the intestinal permeability of patients with Crohn's disease in remission. Scand J Gastroenterol. 2008;43(7):842-8.
15. McFarland LV. Systematic review and meta-analysis of Saccharomyces boulardii in adult patients. World J Gastroenterol. 2010 May 14;16(18):2202-22.
16. Deckelbaum RJ et al. The omega-3 Fatty Acid nutritional landscape: health benefits and sources. J Nutr. 2012 Mar;142(3):587S-91S. Epub 2012 Feb 8.
17. Iwami D et al. Immunomodulatory effects of eicosapentaenoic acid through induction of regulatory T cells. Int Immunopharmacol. 2011 Mar;11(3):384-9.
18. Almallah YZ et al. Distal procto-colitis, natural cytotoxicity, and essential fatty acids. Am J Gastroenterol. 1998 May;93(5):804-9.
19. Hillier K et al. Incorporation of fatty acids from fish oil and olive oil into colonic mucosal lipids and effects upon eicosanoid synthesis in inflammatory bowel disease. Gut 1991;32(10):1151-5.
20. Steinhart AH et al. Nutrition in inflammatory bowel disease. Curr Opinion Gastroenterol. 1997;13(2):140-5.
21. Golias C et al. Physiology and pathophysiology of selectins, integrins, and IgSF cell adhesion molecules focusing on inflammation. A paradigm model on infectious endocarditis. ll Commun Adhes. 2011 Jun;18(3):19-32.
22. Ibrahim A et al. Dietary α-linolenic acid-rich formula reduces adhesion molecules in rats with experimental colitis. Nutrition. 2012 Jan 18.
23. Uchiyama K et al. N-3 polyunsaturated fatty acid diet therapy for patients with inflammatory bowel disease. Inflamm Bowel Dis. 2010 Oct;16(10):1696-707.
24. Stenson WF et al. Dietary supplementation with fish oil in ulcerative colitis. Ann Intern Med. 1992 Apr 15;116(8):609-14.
25. Aslan A et al. Fish oil fatty acid supplementation in active ulcerative colitis: a double-blind, placebo-controlled, crossover study. Am J Gastroenterol. 1992 Apr;87(4):432-7.
26. Wiese DM et al. The effects of an oral supplement enriched with fish oil, prebiotics, and antioxidants on nutrition status in Crohn's disease patients. Nutr Clin Pract. 2011 Aug;26(4):463-73.
27. Hawthorne AB et al. Treatment of ulcerative colitis with fish oil supplementation: a prospective 12 month randomised controlled trial. Gut 1992;33(7):922-8.
28. Belluzzi A et al. Effect of enteric-coated fish-oil preparation on relapses in Crohn's disease. N Engl J Med 1996;334(24):1557-60.
29. Simopoulos AP. Importance of the omega-6/omega-3 balance in health and disease: evolutionary aspects of diet. World Rev Nutr Diet. 2011;102:10-21.
30. Simopoulos AP. The importance of the ratio of omega-6/omega-3 essential fatty acids. Biomed Pharmacother. 2002 Oct;56(8):365-79.
31. Chambers ES et al. The impact of vitamin D on regulatory T cells. Curr Allergy Asthma Rep. 2011 Feb;11(1):29-36.
32. Ooi JH et al. Vitamin D regulation of immune function in the gut: why do T cells have vitamin D receptors? Mol Aspects Med. 2012 Feb;33(1):77-82.
33. Jahnsen J et al. Vitamin D status, parathyroid hormone and bone mineral density in patients with inflammatory bowel disease. Scand J Gastroenterol 2002;37(2):192-9.
34. Wang TT et al. Direct and indirect induction by 1,25-dihydroxyvitamin D3 of the NOD2/CARD15-defensin beta-2 innate immune pathway defective in Crohn disease. J Biol Chem 2010;285(4):2227-31.
35. Lim WC et al. Mechanisms of disease: vitamin D and inflammatory bowel disease. Nat Clin Pract Gastroenterol Hepatol 2005;2(7):308-15
36. Miheller P et al. Comparison of the effects of 1,25 dihydroxyvitamin D and 25 hydroxyvitamin D on bone pathology and disease activity in Crohn's disease patients. Inflamm Bowel Dis 2009;15:1656-1662.
37. Jorgensen SP et al. Clinical trial: Vitamin D3 treatment in Crohn's disease--A randomized double-blind placebo-controlled study. Aliment Pharmacol Ther 2010;32:377-83.
38. Abitbol V et al. Osteoporosis in inflammatory bowel disease: effect of calcium and vitamin D with or without fluoride. Aliment Pharmacol Ther. 2002 May;16(5):919-27.
39. Almeiner HA et al. Oxidative stress and inflammatory bowel disease. Front Biosci 2012;E4:1335-44.
40. Koutroubakis IE et al. Decreased total and corrected antioxidant capacity in patients with inflammatory bowel disease. Dig Dis Sci. 2004 Sep;49(9):1433-7.
41. Kruidenier L et al. Intestinal oxidative damage in inflammatory bowel disease: semi-quantification, localization, and association with mucosal antioxidants. J Pathol. 2003 Sep;201(1):28-36.
42. Trebble TM et al. Fish oil and antioxidants alter the composition and function of circulating mononuclear cells in Crohn disease. Am J Clin Nutr. 2004 Nov;80(5):1137-44.
43. Trebble TM et al. High-dose fish oil and antioxidants in Crohn's disease and the response of bone turnover: a randomised controlled trial. Br J Nutr. 2005 Aug;94(2):253-61.
44. Hengstermann S et al. Altered status of antioxidant vitamins and fatty acids in patients with inactive inflammatory bowel disease. Clin Nutr. 2008 Aug;27(4):571-8.
45. Babickova J et al. Pathological and therapeutic interactions between bacteriophages, microbes and the host in inflammatory bowel disease. World journal of gastroenterology: WJG. Oct 28 2015;21(40):11321-11330.
46. McCarville JL et al. Novel perspectives on therapeutic modulation of the gut microbiota. Therapeutic advances in gastroenterology.Jul 2016;9(4):580-593.
47. Clokie MRJ et al. Phages in nature. Bacteriophage.Jan-Feb 2011;1(1):31-45.
48. Belizario JE et al. Human microbiomes and their roles in dysbiosis, common diseases, and novel therapeutic approaches. Front Microbiol.2015;6:1050.
49. Norman JM et al. Disease-specific alterations in the enteric virome in inflammatory bowel disease. Cell.Jan 29 2015;160(3):447-460.
50. Wang W et al. Metagenomic Analysis of Microbiome in Colon Tissue from Subjects with Inflammatory Bowel Diseases Reveals Interplay of Viruses and Bacteria. Inflamm Bowel Dis.Jun 2015;21(6):1419-1427.
51. Loc-Carrillo C et al. Pros and cons of phage therapy. Bacteriophage.Mar-Apr 2011;1(2):111-114.
52. Bruttin A et al. Human volunteers receiving Escherichia coli phage T4 orally: a safety test of phage therapy. Antimicrobial agents and chemotherapy.Jul 2005;49(7):2874-2878.
53. Sarker SA et al. Oral T4-like phage cocktail application to healthy adult volunteers from Bangladesh. Virology.Dec 20 2012;434(2):222-232.
54. Taylor RA et al. Curcumin for inflammatory bowel disease: a review of human studies. Altern Med Rev. 2011 Jun;16(2):152-6.
55. Holt PR et al. Curcumin therapy in inflammatory bowel disease: a pilot study. Dig Dis Sci. 2005 Nov;50(11):2191-3.
56. Hanai H et al. Curcumin maintenance therapy for ulcerative colitis: Randomized, multicenter, double-blind, placebo-controlled trial. Clin Gastroenterol Hepatol 2006;4(12):1502-6.
57. Gerhardt H et al. Therapy of active Crohn disease with Boswellia serrata extract H 15. Z Gastroenterol 2001;39:11-7 [in German].
58. Gupta I et al. Effects of gum resin of Boswellia serrata in patients with chronic colitis. Planta Med 2001;67:391–5.
59. Gupta I et al. Effects of Boswellia serrata gum resin in patients with ulcerative colitis. Eur J Med Res 1997;2:37-43.
60. Holtmeier W et al. Randomized, placebo-controlled, double-blind trial of Boswellia serrata in maintaining remission of Crohn's disease: good safety profile but lack of efficacy. Inflamm Bowel Dis 2011;17(2):573-82.
61. Sengupta K et al. Cellular and molecular mechanisms of anti-inflammatory effect of Aflapin: a novel Boswellia serrata extract. Mol Cell Biochem. 2011 Aug;354(1-2):189-97. Epub 2011 Apr 11.
62. Vieira EL et al. Oral administration of sodium butyrate attenuates inflammation and mucosal lesion in experimental acute ulcerative colitis. J Nutr Biochem. 12 Jun 2011, 23(5):430-436
63. Segain JP et al. Butyrate inhibits inflammatory responses through NFkappaB inhibition: implications for Crohn's disease. Gut. 2000 Sep;47(3):397-403.
64. Assisi RF; GISDI Study Group. Combined butyric acid/mesalazine treatment in ulcerative colitis with mild-moderate activity. Results of a multicentre pilot study. Minerva Gastroenterol Dietol 2008;54(3):231-8.
65. Di Sabatino A et al. Oral butyrate for mildly to moderately active Crohn's disease. Aliment Pharmacol Ther 2005;22(9):789-94.
66. Langmead L et al. Randomized, double-blind, placebo-controlled trial of oral aloe vera gel for active ulcerative colitis. Aliment Pharmacol Ther. 2004a Apr 1;19(7):739-47.
67. Langmead L et al. Anti-inflammatory effects of aloe vera gel in human colorectal mucosa in vitro. Aliment Pharmacol Ther. 2004b Mar 1;19(5):521-7.
68. Omer B et al. Steroid-sparing effect of wormwood (Artemisia absinthium) in Crohn's disease: A double-blind placebo-controlled study. Phytomedicine 2007;14(2-3):87-95.
69. Krebs S et al. Wormwood (Artemisia absinthium) suppresses tumour necrosis factor alpha and accelerates healing in patients with Crohn's disease - A controlled clinical trial. Phytomedicine. 2010 Apr;17(5):305-9.
70. Geerling BJ et al. Comprehensive nutritional status in recently diagnosed patients with inflammatory bowel disease compared with population controls. Eur J Clin Nutr 2000a;54(6):514-21.
71. Hinks IJ et al. Reduced concentration of selenium in mild Crohn's disease. J Clin Pathol 1988;41:198-201.
72. Ojuawo A et al. The serum concentrations of zinc, copper and selenium in children with inflammatory bowel disease. Cent Afr J Med. 2002 Sep-Oct;48(9-10):116-9.
73. Geerling BJ et al. Nutritional supplementation with N-3 fatty acids and antioxidants in patients with Crohn's disease in remission: effects on antioxidant status and fatty acid profile. Inflamm Bowel Dis 2000b;6(2):77-84.
74. Abd-Allah AR et al. Pro-inflammatory and oxidative stress pathways which compromise sperm motility and survival may be altered by L-carnitine. Oxid Med Cell Longev. 2009 Apr-Jun;2(2):73-81.
75. Buyse J et al. Dietary L-carnitine supplementation enhances the lipopolysaccharide-induced acute phase protein response in broiler chickens. Vet Immunol Immunopathol. 2007 Jul 15;118(1-2):154-9.
76. Shakeri A et al. Effects of L-carnitine supplement on serum inflammatory cytokines, C-reactive protein, lipoprotein (a), and oxidative stress in hemodialysis patients with Lp (a) hyperlipoproteinemia. Hemodial Int. 2010 Oct;14(4):498-504.
77. Yuan Y et al. Protective effects of L-carnitine on intestinal ischemia/reperfusion injury in a rat model. J Clin Med Res. 2011 Apr 4;3(2):78-84
78. Mikhailova TL et al. Randomised clinical trial: the efficacy and safety of propionyl-L-carnitine therapy in patients with ulcerative colitis receiving stable oral treatment. Aliment Pharmacol Ther. 2011 Nov;34(9):1088-97.
79. Den Hond E et al. Effect of long-term oral glutamine supplements on small intestinal permeability in patients with Crohn's disease. JPEN J Parenter Enteral Nutr 1999;23(1):7-11.
80. Kruschewski M et al. [Protective effect of glutamine on microcirculation of the intestine in experimental colitis]. Langenbecks Arch Chir Suppl Kongressbd. 1998;115(Suppl I):229-31.
81. Sido B et al. Low intestinal glutamine level and low glutaminase activity in Crohn's disease: a rational for glutamine supplementation? Dig Dis Sci 2006;51(12):2170-9.
82. Benjamin J et al. Glutamine and Whey Protein Improve Intestinal Permeability and Morphology in Patients with Crohn's Disease: A Randomized Controlled Trial. Dig Dis Sci. 2011 Oct 26.
83. Ockenga J et al. Glutamine-enriched total parenteral nutrition in patients with inflammatory bowel disease. Eur J Clin Nutr. 2005 Nov;59(11):1302-9.
84. Krasinski SD, Russell RM et al. The prevalence of vitamin K deficiency in chronic gastrointestinal disorders. Am J Clin Nutr 1985;41(3):639-43.
85. Nakajima S et al. Association of vitamin K deficiency with bone metabolism and clinical disease activity in inflammatory bowel disease. Nutrition 2011;27(10):1023-8.
86. Duggan P et al. Vitamin K status in patients with Crohn's disease and relationship to bone turnover. Am J Gastroenterol 2004;99(11):2178-85.
87. Hou JK et al. Dietary intake and risk of developing inflammatory bowel disease: a systematic review of the literature. Am J Gastroenterol. 2011 Apr;106(4):563-73.
88. Sakamoto N et al. Dietary risk factors for inflammatory bowel disease: a multicenter case-control study in Japan. Inflamm Bowel Dis. 2005 Feb;11(2):154-63.
89. Heaton KW et al. Treatment of Crohn's disease with an unrefined-carbohydrate, fiber-rich diet. Br Med J 1979;ii:764-6.
90. Hanai H, Kanauchi O, et al. Germinated barley foodstuff prolongs remission in patients with ulcerative colitis. Int J Mol Med 2004;13(5):643-7.
91. Bubenik GA. Gastrointestinal melatonin: localization, function, and clinical relevance. Dig Dis Sci 2002;47(10):2336-48.
92. Johe PD et al. The in vivo effect of melatonin on cellular activation processes in human blood during strenuous physical exercise. J Pineal Res 2005;39:324–330.
93. Terry PD et al. Melatonin and ulcerative colitis: evidence, biological mechanisms, and future research. Inflamm Bowel Dis 2009;15:134-40.
94. Boznanska P et al. 24-hour urinary 6-hydroxymelatonin sulfate excretion in patients with ulcerative colitis. Pol Merkur Lekarski 2007;22(131):369-72 [in Polish].
95. Chojnacki C et al. Evaluation of melatonin effectiveness in the adjuvant treatment of ulcerative colitis. J Physiol Pharmacol 2011;62(3):327-34.
96. Rakhimova OIu. Use of melatonin in combined treatment for inflammatory bowel diseases. Ter Arkh 2010;82(12):64-8 [in Russian].
97. Maldonado MD et al. Melatonin usage in ulcerative colitis: a case report. J Pineal Res 2008;45(3):339-40.
98. Haden ST et al. Effects of age on serum dehydroepiandrosterone sulfate, IGF-I, and IL-6 levels in women. Calcif Tissue Int. 2000 Jun;66(6):414-8.
99. Head KA et al. Inflammatory bowel disease Part 1: ulcerative colitis--pathophysiology and conventional and alternative treatment options. Altern Med Rev. 2003 Aug;8(3):247-83.
100. Andus T et al. Patients with refractory Crohn's disease or ulcerative colitis respond to dehydroepiandrosterone: a pilot study. Aliment Pharmacol Ther. 2003 Feb;17(3):409-14.
101. Straub RH et al. Replacement therapy with DHEA plus corticosteroids in patients with chronic inflammatory diseases: substitutes of adrenal and sex hormones. Z Rheumatol. 2000;59(suppl 2):II/108-18.
102. Alastair F et al. Nutrition in inflammatory bowel disease. JPEN J Parenter Enteral Nutr. 2011 Sep;35(5):571-80.
103. Mortimore M et al. A role for B₁₂ in inflammatory bowel disease patients with suppurative dermatoses? An experience with high dose vitamin B₁₂ therapy. J Crohns Colitis. 2010 Oct;4(4):466-70.
104. Campos FG et al. Pharmacological nutrition in inflammatory bowel diseases. Nutr Hosp. 2003 Mar;18(2):57-64.
105. Oussalah A et al. ta-analysis: hyperhomocysteinaemia in inflammatory bowel diseases. Aliment Pharmacol Ther. 2011 Nov;34(10):1173-84.
106. Roblin X et al. Factors associated with hyperhomocysteinemia in inflammatory bowel disease: prospective study in 81 patients [in French]. Rev Med Interne. 2006 Feb;27(2):106-10.
107. Zezos P et al. Hyperhomocysteinemia in ulcerative colitis is related to folate levels. World J Gastroenterol. 2005 Oct 14;11(38):6038-42.
108. Mahmood A et al. Prevalence of hyperhomocysteinaemia, activated protein C resistance and prothrombin gene mutation in inflammatory bowel disease. Eur J Gastroenterol Hepatol 2005;17:739-44.
109. Fernandez-Miranda C et al. Hyperhomocysteinemia and methylenetetrahydrofolate reductase 677C-->T and 1298A-->C mutations in patients with inflammatory bowel disease. Rev Esp Enferm Dig. 2005 Jul;97(7):497-504.
110. Srirajaskanthan R et al. Venous thrombosis in inflammatory bowel disease. Eur J Gastroenterol Hepatol. 2005 Jul;17(7):697-700.
111. Papa A et al. Increased carotid intima-media thickness in patients with inflammatory bowel disease. Aliment Pharmacol Ther. 2005 Nov 1;22(9):839-46.
112. Patel V et al. Methotrexate for maintenance of remission in Crohn's disease. Cochrane Database Syst Rev 2009 Oct 7;(4):CD006884.
113. Zintzaras E. Genetic variants of homocysteine/folate metabolism pathway and risk of inflammatory bowel disease: a synopsis and meta-analysis of genetic association studies. Biomarkers. 2010 Feb;15(1):69-79.
114. Etzel JP et al. Assessment and management of low bone density in inflammatory bowel disease and performance of professional society guidelines. Inflamm Bowel Dis. 2011 Jan 13.
115. Harpavat M et al. Metabolic bone disease in inflammatory bowel disease. J Clin Gastroenterol. 2004 Mar;38(3):218-24.
116. Miheller P et al. Clinical relevance of changes in bone metabolism in inflammatory bowel disease. World J Gastroenterol. 2010 Nov 28;16(44):5536-42.
117. Agrawal M et al. Bone, inflammation, and inflammatory bowel disease. Curr Osteoporos Rep. 2011 Dec;9(4):251-7.
118. Kuwabara A et al. High prevalence of vitamin K and D deficiency and decreased BMD in inflammatory bowel disease. Osteoporos Int 2009;20(6):935-42.
119. Rogler G et al. Extraintestinal manifestations of inflammatory bowel disease [in German]. Med Klin (Munich). 2004 Mar 15;99(3):123-30.
120. Bernstein CN et al. The association between corticosteroid use and development of fractures among IBD patients in a population-based database. Am J Gastroenterol 2003;98(8):1797-801.
121. Kappelman MD et al. Thromboembolic risk among Danish children and adults with inflammatory bowel diseases: a population-based nationwide study. Gut 2011;60:937–43.
122. Solem CA et al. Venous thromboembolism in inflammatory bowel disease. Am J Gastroenterol 2004;99(1):97-101.
123. Sonoda K et al. Evaluation of venous thromboembolism and coagulation-fibrinolysis markers in Japanese patients with inflammatory bowel disease. J Gastroenterol 2004;39(10):948-54.
124. Koutroubakis IE. Therapy insight: Vascular complications in patients with inflammatory bowel disease. Nat Clin Pract Gastroenterol Hepatol 2005;2(6):266-72.
125. Phang M et al. Diet and thrombosis risk: nutrients for prevention of thrombotic disease. Semin Thromb Hemost 2011;37(3):199-208.
126. Mitamura T et al. The more an ulcerative colitis is repeated, the more the risk of colorectal carcinogenesis is increased in mice. Anticancer Res. 2002 Nov-Dec;22(6C):3955-61.
127. Itzkowitz SH et al. Inflammation and cancer IV. Colorectal cancer in inflammatory bowel disease: the role of inflammation. Am J Physiol Gastrointest Liver Physiol. 2004 Jul;287(1):G7-G17.
128. Phelip JM et al. Association of hyperhomocysteinemia and folate deficiency with colon tumors in patients with inflammatory bowel disease. Inflamm Bowel Dis. 2008 Feb;14(2):242-8.
129. Kim DH et al. Pooled analyses of 13 prospective cohort studies on folate intake and colon cancer. Cancer Causes Control. 2010 Nov;21(11):1919-30. Epub 2010 Sep 5.
130. Biasco G et al. Folate and prevention of colorectal cancer in ulcerative colitis. Eur J Cancer Prev. 2005 Aug;14(4):395-8.
131. Fife J et al. Folic acid supplementation and colorectal cancer risk: a meta-analysis. Colorectal Dis. 2011 Feb;13(2):132-7.
132. Yakut M et al. Serum vitamin B12 and folate status in patients with inflammatory bowel diseases. Eur J Intern Med. 2010 Aug;21(4):320-3.
参考来源:
美国国立公众健康网
www.medlineplus.gov
美国胃肠病协会
http://www.gastro.org
美国家庭医生学会
http://www.familydoctor.org
美国国立糖尿病、消化和肾病研究所
http://www.niddk.nih.gov
加拿大胃肠病协会
https://www.cag-acg.org
加拿大卫生部
http://www.hc-sc.gc.ca
免责声明和安全信息
- 本信息(包括任何附带资料)不是为了取代医生或有关合格从业人士的建议或忠告。
- 任何人如果想要对本文涉及的药物、饮食、运动或其他生活方式的使用、或改变调整,以预防或治疗某一特定健康状况或疾病,应首先咨询医生或有关合格从业人士,并获得他/她们的许可。妊娠和哺乳妇女在使用本网站任何内容前,尤其应征求医生的意见。
- 除非另有说明,本网站所述内容仅适用于成人。
- 本网站所推荐的任何产品,消费者应该以实际的产品标签内容为准,尤其应关注重要的安全信息以及产品最新信息,包括剂量、使用方法和禁忌症等。
- 由于循证医学研究、文献及有关产品处于不断的变化中,本网站工作人员将尽力更新。
- 本网站不能保证所载文章内容、综合干预方案以及相关成分或产品述及的健康益处,也不承担任何责任。

